
HOME
Hello! Welcome to the chemicals’ page of Yixiao, Miao Ran and Chen Xi!
This is where you can find information on Jurong Island, and two out of the thousands of useful chemicals produced on the island.
Navigations are on your right!
About Jurong Island: Learn about Jurong Island, past and present
Chlorine: Discover the manufacture process and applications of chlorine
Superabsorbent Polymers: Ever wonder what's in a diaper? Click to find out more!
Bibliography: Thanks to these sources that helped us in our work!
Enjoy!
jurong island
AN INTRODUCTION
Known to all, Singapore comprises of one main island and several other small islands, including Pulau Ayer Chawan, Pulau Ayer Merbau, Pulau Merlimau etc, of which Sentosa is the most commercially developed. The other islands were underdeveloped and basically the land was left barren for years.
In the late 1960s and early 1970s, three of the biggest oil companies in te world decided to house their facilities on the islands - Esso in Pulau Ayer Chawan, Singapore Refinery Company in Pulau Merlimau and Mobil Oil in Pulau Pesek. These three companies then became the firsts of Singapore’s petroleum industry.
In the 1980s, Singapore identified the chemicals sector as an important cluster for the country’s economic growth, this was an industry which would contribute significantly. Seven islands was then envisaged to become Singapore’s petrochemicals hub in what was called the Jurong Island Project. Industrial land was becoming scarce on Singapore mainland, thus the idea of joining these 3 southern islands off Jurong to form one colossal island to create more industrial land for industrialization.
In 1991, JTC was put in charge for the development of Jurong Island. After investigating the needs of the industrialists hoping to set up on the to-be Jurong-island, and gathering their feedback and suggestions, JTC planned and corresponded with government agencies to deliver infrastructure and services to the island, in terms of roads, drains, utilities and others.
Physical land reclamation began in 1995, and Jurong Island was officially opened in October 2000. From the 991-hectare land area of the original seven islets, Jurong Island currently has a total land area of approximately 3200 hectares.
Since then, Jurong Island has been transformed from a sporadic collection of seven islets into an integrated island of industrial excellence with only one aim in mind: to nurture an optimal concentration of fully integrated chemical and supporting industries. The vision for Jurong Island is to develop it into a world-class chemicals hub in the Asia-Pacific region.
Enjoying a strategic geographical location at the heart of the Association of South-East Asian Nations (ASEAN), well served by air, sea and telecoms connectivity, Singapore and Jurong Island are only seven hours away from any major Asian destination. This makes Jurong Island an excellent place for investments and a strategic launch pad to reach key markets in the Asia-Pacific region. Its strategic location also makes it an optimum point to distribute goods worldwide. Singapore is also home to the world’s busiest port. Its excellent port infrastructure is highly valued by many investors. Propelled by the combination of many success factors, Jurong island is being nurtured to be the leading symbol of Singapore’s National enterprise.
CHLORINE
MANUFACTURE PROCESS
Did you know that chlorine can be manufactured by electrolysis? All you have to do is mix sodium chloride solution,
or commonly known as brine, with water, place the mixture in membrane electrolytic cells and tada - You get CHLORINE!
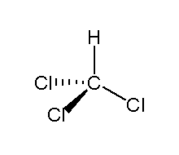
Actually, the production of chlorine also results in the co-products caustic soda, sodium hydroxide, and hydrogen gas. The equation
of the reaction is given as:
2 NaCl (aq) + 2 H2O (l) → H2 (g) + Cl2 (g) + 2 NaOH (aq)
Sodium chloride + water → hydrogen gas + chlorine gas + sodium hydroxide
To understand what “membrane electrolytic cells” is, we must first understand the term “electrolysis”. Electrolysis is a method of
separating bonded elements and compounds by passing an electric current through them. It is an efficient method of producing new chemicals
from reactants.
Now we’re ready to explore how chlorine is manufactured though membrane electrolytic cells.
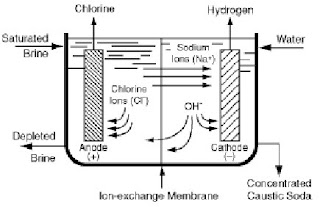
Firstly, sodium chloride is fed to the anode compartment (negative side of the cell). When chlorine is liberated at the anode, it dissolves in water and dissociates (split) into sodium cations (Na+) and chloride anions (Cl –).
At the anode, the chloride ions lose electrons to form chlorine gas while at the cathode, water molecules gain electrons to form hydrogen gas. Chloride is said to be oxidized and water reduced. The sodium ions (Na+) in the solution and the hydroxyl ions (OH–) produced at the cathode constitute the components of sodium hydroxide formed during the electrolysis of sodium chloride.
Cathode: 2 H+ (aq) + 2 e – → H2 (g)
Anode: 2 Cl – → Cl2 (g) + 2 e –
In membrane cells, an ion-exchange membrane which is a bi-layer membrane made of perfluorocarboxylic and perfluorosulfonic acid-based films, is used as a separator to divide the anode and cathode compartments. This prevents the mixing of chlorine gas and the sodium hydroxide in order to ensure generation of pure products. When sodium chloride is added, only the sodium ions and some water migrate through the membrane. The unreacted sodium chloride and other inert ions remain in the anolyte.
The chlorine gas from the anode compartment contains moisture and some hydrogen. It is first cooled to 60oF and passed through demisters to remove the water droplets and the particulates of salt. Demister is a device that removes liquid droplets from a gas stream. The cooled gas goes to sulfuric acid circulating towers where the moisture is removed. The dried chlorine then goes through demisters again. This gas is used for producing hypochlorite or hydrochloric acid. Alternatively, it can be
neutralized (pH 7) with caustic soda or lime (calcium hydroxide) to form hypochlorite which is sold as bleach or decomposed to form salt and oxygen.
The chlorine gas from the anode compartment contains moisture and some hydrogen. It is first cooled to 60oF and passed through demisters to remove the water droplets and the particulates of salt. The cooled gas goes to sulfuric acid circulating towers where the moisture is removed. The dried chlorine then goes through demisters again. This gas is used for producing hypochlorite or hydrochloric acid. Alternatively, it can be
neutralized with caustic soda or lime (calcium hydroxide) to form hypochlorite which is sold as bleach or decomposed to form salt and oxygen.
APPLICATIONS
Chlorine, being very reactive, forms compounds with other substances rapidly.
Below is a video showing the reaction between sodium and chlorine.
This property of chlorine allows many useful chlorine compounds to be formed. In the medical field, chlorine also plays a crucial role as it is the component of majority of the medicines or the medicine are developed using products which contain chlorine. For example, a type of anaesthetic used during surgery is chloroform (CHCl3 ).

Other examples of daily-products which contain chlorine are glue,
paints, solvents, foam rubbers,
car bumpers, food additives, pesticides and antifreeze. One of the most commonly used chlorine-containing substances is
PVC (poly vinyl chloride).
PVC is widely used, for example in drainpipes, insulation wires, floors, windows,
bottles and waterproof clothes.
Other than this, chlorine also has the ability to form bonds between two substances that do not normally react with one another. For example, chlorine can bond to substances that contain carbon atoms, forming organic substances such as plastic, solvents and oils, and several human body fluids.
Chlorine-based bleach is applied as a disinfectant very effective for the deactivation of pathogenic microorganisms (bacteria and viruses) in drinking water, swimming pool water and wastewater. It does so by breaking the chemical bonds in their molecules. Disinfectants that are used for this purpose consist of chlorine compounds which can exchange atoms with other compounds, such as enzymes in bacteria and other cells. When enzymes contacts with chlorine, one or more hydrogen atoms in the molecule are replaced by chlorine. This causes the entire molecule to change shape or fall apart. When enzymes cease to function, a cell or bacterium will die.
In addition to this, chlorine can be also used to bleach things like clothes and paper. Chlorine’s ‘bleaching effect’ is due to chlorine or hypochlorite oxidation. Bleach is actually chlorine gas dissolved in an alkali-solution, such as sodium hydroxide (NaOH). When chlorine is dissolved in an alkaline solution, hypochlorite ions (OCl-) are formed during an autoredox reaction Cl2 + H2O -> HOCl + H+ + Cl- . For example, chlorine reacts with sodium hydroxide to sodium hypochlorite (NaOCl). This is a very good disinfectant with a stable effect. Bleaching powder (CaOCl2) is another alternative, produced by passing chlorine through calcium hydroxide (CaOH).
SUPERABSORBENT POLYMERS
MANUFACTURE PROCESS
Super absorbent polymers are cross-linked networks of flexible polymer chains. A “polymer” is a substance composed of molecules with large molecular mass composed of repeating structural units connected by covalent chemical bonds. A “polymer chain” is the simplest form of polymer molecule composed of a single main chain (straight chain or linear polymer).

The above diagram shows the structure of Polyacrylic acid which contains an ionizable group on each repeat unit (-COOH).
Super absorbent powder polymer production begins with solution polymerization. Polymerization is a process of reacting monomer molecules together in a chemical reaction to form three-dimensional networks of polymer chains.

In solution polymerization, partially neutralized acrylic acid, blended with sodium hydroxide in the presents of an initiator (cross-linking agent), forms a sodium salt polymer of polyacrylic acid.
Acrylic acid + sodium hydroxide +water → sodium salt polymer

The polymerization results in a water insoluble, water swellable gel containing approximately 25 to 40 % solids. These gel granules are passed to the drying process and surface coating to enhance the property before drying and packing. Then these granules are sifted to produce a powdered SAP product with a typical particle size ranging between 100 to 800 µm. The finished SAP product is then shipped to a hygiene-industry converter where it is blended with fibrillated wood fluff to form the absorbent core structure of a personal hygiene article such as a diaper.

APPLICATIONS
One important property of super absorbent polymers is that it retains water – some will absorb up to 1,000 g of water per gram of polymer. The largest use of super absorbent polymers is therefore in the hygiene-industry. These consumer products include, in order of volume of super absorbents used, disposable infant diapers, children’s training pants, adult incontinence articles, and feminine sanitary napkins.
The most important characteristic in a super absorbent used in the personal care market is the extent of swelling. The total absorbency and swelling capacity are controlled by the type and degree of cross-linking to the polymer. A cross-link suggests a branch point from which four or more distinct chains emanate (originate).

The mechanism of swelling of ionized, cross-linked polymer networks is based upon the concept of osmotic pressure. The polymer acts as a semi-permeable membrane which does not allow charged substituents to exit the polymer into the surrounding solution. This is because the ionized units contain fixed charges which attract and fix ions from the outer solution. Therefore, a charge gradient is set up, in which the concentration of free ions is greater outside of the polymer. Therefore, the osmotic pressure exerted by the gradient causes the polymer chain to swell as further ions diffuse in. If the super absorbent polymer is more highly cross-linked, it is more rigid in the swollen state. By improving the rigidity of the particles, it enables the swelling particles to push aside the fiber component of the composite, thereby maintaining the porosity and permeability during subsequent contacts with liquid, therefore allowing for better absorption.
Thus, low density cross-linked SAPs (less chains at one branch point) have higher absorbent capacities and swell to a larger degree. These products also have a softer and more cohesive type gel formation. High cross-link density polymers (more chains at one branch point) exhibit lower absorbent capacity and swell. The gel strength is firmer and can maintain particle shape even under modest pressure. This high density cross-linked SAPs are therefore used in diapers.

Super absorbent polymer is added to baby diapers using the blended application. The super absorbent polymer first is mixed homogeneously with the fluff pulp. Then the mixture is laid down to give the absorbent structure, which is subsequently covered with a non-woven fabric. Super absorbent polymers absorb, and retain under a slight mechanical pressure, about 30 times their weight in urine. The swollen gel holds the liquid in a solid, rubbery state and prevents the liquid from leaking onto the baby’s skin and clothing. These properties of the super absorbent polymer make it suitable for production of personal hygiene products such as diaper.
Another feature that makes SAPs useful in the personal hygiene industry is their ability to respond strongly to very weak external stimuli, such as minute changes in pH or temperature. For example, a polymer gel might first absorb a quantity of liquid and later release it as the external conditions change. Timed release of pharmaceuticals is one example among many in which a control stimulus determines the rate of release. Cross-linking in the polymer microstructure is again a key player here that governs actual performance of the product.
For similar reasons, super absorbent polymers are also used for blocking water ingress in underground cable, horticultural water retention agents, control of spill and waste fluid (aqueous), artificial snow for motion picture and stage production.
Super absorbent polymers offer the absorbency of granular polymers supplied in solution form. In this case, solutions are diluted with water prior to application. It is then used to coat onto substrates. After drying at a specific temperature for a specific time, the result is a coated substrate with super absorbent functionality. This chemistry can be applied directly onto wires & cables, though it is especially optimized for use on components such as rolled goods or sheeted substrates.
CONCLUSION
So, that is all for our tour of Jurong Island and the short lesson on the two chemicals’ applications today! We hope that you have learnt more about the operations of one of Singapore’s leading industries, and of course about the chemicals. Now if your parents happen to test you about how diapers are made, you’ve got something to boast about!
Thank you for visiting our website and have a good day!
BIBLIOGRAPHY
DIAGRAMS
http://www.vinythai.co.th/ourchemicalproducts/causticsoda/mkt1chemicalindustry/app1superabsorbent/0,,2187-2-0,00.htm
http://nexant.ecnext.com/coms2/gi_0255-3047/Super-Absorbent-Polymers-SAP.html
www.functionalpolymers.basf.com/portal/streamer?fid=291076
http://electrochem.cwru.edu/ed/encycl/art-b01-brine.htm
INFORMATION
http://www.eng.buffalo.edu/Courses/ce435/Diapers/Diapers.html
http://en.wikipedia.org/wiki/Superabsorbent_polymer
http://composite.about.com/library/glossary/s/bldef-s5018.htm
http://concise.britannica.com/ebc/art-272
http://www.sciencedirect.com/science?_ob=ArticleURL&_udi=B6TWW-4846DRN-1&_user=10&_coverDate=07%2F31%2F2003&_rdoc=1&_fmt=&_orig=search&_sort=d&view=c&_acct=C000050221&_version=1&_urlVersion=0&_userid=10&md5=902f40ba3e3f569e7662ab7a6168c69b
http://www.vinythai.co.th/ourchemicalproducts/causticsoda/mkt1chemicalindustry/app1superabsorbent/0,,2187-2-0,00.htm
http://nexant.ecnext.com/coms2/gi_0255-3047/Super-Absorbent-Polymers-SAP.html
http://electrochem.cwru.edu/ed/encycl/art-b01-brine.htm
http://www.ercoworldwide.com/products_chlorine.asp
http://www.lenntech.com/water-disinfection/disinfectants-chlorine.htm
www.functionalpolymers.basf.com/portal/streamer?fid=291076

 Firstly, sodium chloride is fed to the anode compartment (negative side of the cell). When chlorine is liberated at the anode, it dissolves in water and dissociates (split) into sodium cations (Na+) and chloride anions (Cl –).
At the anode, the chloride ions lose electrons to form chlorine gas while at the cathode, water molecules gain electrons to form hydrogen gas. Chloride is said to be oxidized and water reduced. The sodium ions (Na+) in the solution and the hydroxyl ions (OH–) produced at the cathode constitute the components of sodium hydroxide formed during the electrolysis of sodium chloride.
Cathode: 2 H+ (aq) + 2 e – → H2 (g)
Firstly, sodium chloride is fed to the anode compartment (negative side of the cell). When chlorine is liberated at the anode, it dissolves in water and dissociates (split) into sodium cations (Na+) and chloride anions (Cl –).
At the anode, the chloride ions lose electrons to form chlorine gas while at the cathode, water molecules gain electrons to form hydrogen gas. Chloride is said to be oxidized and water reduced. The sodium ions (Na+) in the solution and the hydroxyl ions (OH–) produced at the cathode constitute the components of sodium hydroxide formed during the electrolysis of sodium chloride.
Cathode: 2 H+ (aq) + 2 e – → H2 (g)







